Pharmacogenetic Testing: BioCertica vs Traditional Methods
BioCertica Content TeamThe field of pharmacogenetic testing is still relatively new and requires much progress. As you may have read, in pharmacogenomic testing, DNA is genotyped and matched to known gene-drug interactions; however, there is no standardized approach for reporting this testing. This leads to many differences in how we compile and present these reports. Therefore BioCertica’s approach to presenting pharmacogenetic results is different from other companies that offer this testing in South Africa. For the sake of transparency, we would like to explain the difference.
Pharmacogenetic testing of our competitors
We have noticed that our competitors tend to follow a similar approach.
For their pharmacogenomic tests, our competitor starts with the gene-drug interactions. They look at a few gene-drug interactions with a specific focus on the CYP genes, a group of enzymes that metabolize many drugs. Specific phenotypes like poor, intermediate, normal, and rapid metabolizer associate with these gene-drug interactions. From there, they map these phenotypes to each drug that will be metabolized by those enzymes.

Pharmacogenetic testing at BioCertica
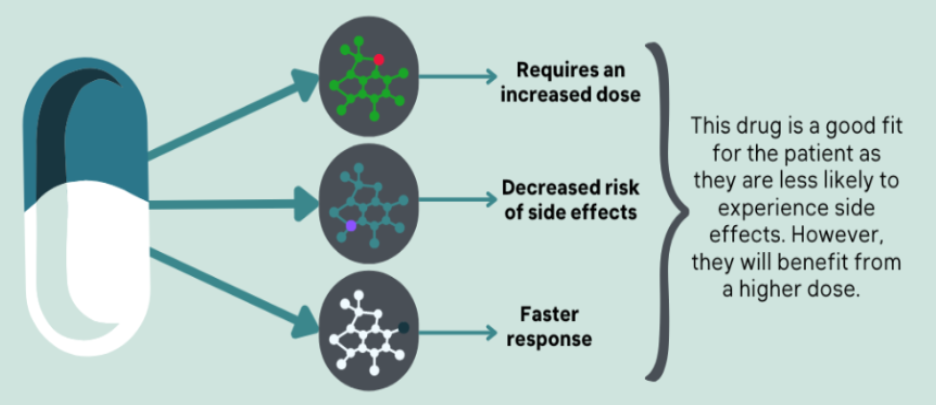
At BioCertica, we effectively report the gene-drug interactions in the opposite direction because instead of starting with the gene-drug interaction, we start with the drug. For each drug in our databases, we look for the known gene-drug interactions associated with the drug of interest. Although CYP enzymes metabolize many drugs, other gene-drug interactions also play a part in drug metabolism, and you can find them on the PharmacoPGK databases. Therefore we can provide information for multiple gene-drug interactions that are specifically related to that drug.
We chose this approach to provide more focused, drug-specific information. Typically, individuals getting a pharmacogenomic test are already on medication and want to know if it's suitable or if they should try an alternative. Therefore we feel that our approach to reporting pharmacogenomic results is more intuitive from a user’s perspective.

Please read our other articles here for more information on BioCertica’s pharmacogenomic tests.
Written by: Jamie Fernandez, B.Sc. Hons. in Genetics, Content Specialist



